Rifampin/Penicillin-Resistant Strain of RB51 Brucella Contracted from Consumption of Raw Milk
Distributed via the CDC Health Alert Network
September 13, 2017, 1230 ET (12:30 PM ET)
CDCHAN-00407
Summary:
The Texas Department of State Health Services, with assistance from CDC, is investigating Brucella RB51 exposures and illnesses that may be connected to the purchase and consumption of raw (unpasteurized) milk from K-Bar Dairy in Paradise, Texas. Symptoms of brucellosis can include: fever, sweats, malaise, anorexia, headache, fatigue, muscle & joint pain, and potentially more serious complications (e.g., swelling of heart, liver, or spleen, neurologic symptoms).
Background
A person who drank raw milk from K-Bar Dairy in Paradise, Texas, has been hospitalized with brucellosis. Milk samples from the dairy have tested positive for a Brucella strain called RB51. People who consumed milk or milk products from this dairy from June 1, 2017, to August 7, 2017 are at an increased risk for brucellosis and should receive appropriate post-exposure prophylaxis (PEP). They are advised to consult with their health care providers regarding PEP care and possible diagnostic testing. Please note: the incubation period for brucellosis can range from five days to six months.
Recommendations
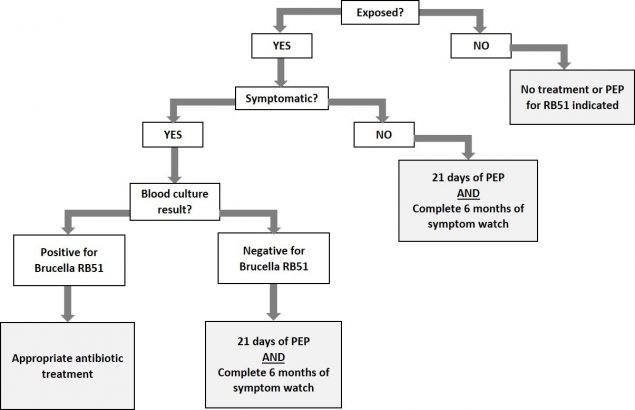
Brucella strain RB51 is resistant to rifampin and penicillin. A combination of doxycycline and trimethoprim/sulfamethoxazole for 21 days is the recommended first-line PEP regimen for RB51 exposure. There is no serological test available to detect RB51 infection. Blood culture is the recommended diagnostic test for exposed symptomatic individuals. When ordering blood cultures to diagnose brucellosis, please advise the laboratory that blood culture may grow Brucella and that appropriate laboratory1 precautions should be observed. If brucellosis occurs despite prophylaxis, treatment regimens2 should be selected based on antimicrobial susceptibility results. Please see the diagram below for information on developing an evaluation and treatment plan for exposed patients. Women who are pregnant or breastfeeding should consult with their health care provider.
from K-Bar Dairy June 1, 2017 – August 7, 2017
For More Information
Risks from Unpasteurized Dairy Products
https://www.cdc.gov/brucellosis/exposure/unpasteurized-dairy-products.html
Exposure to RB51 through Raw Milk or Milk Products: How to Reduce Risk of Infection
https://www.cdc.gov/brucellosis/clinicians/rb51-raw-milk.html
https://www.cdc.gov/brucellosis/symptoms/index.html
Brucellosis and Expecting Mothers
https://www.cdc.gov/brucellosis/exposure/expecting-mothers.html
Raw Milk Questions and Answers
https://www.cdc.gov/foodsafety/rawmilk/raw-milk-questions-and-answers.html
CDC-INFO
https://www.cdc.gov/cdc-info/index.html or 1-800-232-4636
CDC Emergency Operations Center (EOC)
770-488-7100
Bacterial Special Pathogens Branch
bspb@cdc.gov or 404-639-1711
References
1Biosafety in Microbiological and Biomedical Laboratories
https://www.cdc.gov/biosafety/publications/bmbl5/bmbl.pdfCdc-pdf
2Treatment of Brucellosis (for alternative treatment guidance)
- Ariza J et al. 2007. Perspectives for the Treatment of Brucellosis in the 21st Century: The Ioannina Recommendations. PLoS Med. 4(12): e317. http://www.plosmedicine.org/article/info:doi/10.1371/journal.pmed.0040317External
- Al-Tawfiq JA. 2008. Therapeutic options for human brucellosis. Expert Rev Anti Infect Ther. 6(1): 109-120. http://www.ncbi.nlm.nih.gov/pubmed/18251668External
- Solera J. 2010. Update on brucellosis: therapeutic challenges. Intl J Antimicrob Agent. 36S, S18–S20. http://www.ncbi.nlm.nih.gov/pubmed/20692127External
Brucellosis Reference Guide
https://www.cdc.gov/brucellosis/pdf/brucellosi-reference-guide.pdfCdc-pdf
The Centers for Disease Control and Prevention (CDC) protects people’s health and safety by preventing and controlling diseases and injuries; enhances health decisions by providing credible information on critical health issues; and promotes healthy living through strong partnerships with local, national and international organizations.
Department of Health and Human Services
HAN Message Types
- Health Alert: Conveys the highest level of importance; warrants immediate action or attention.
- Health Advisory: Provides important information for a specific incident or situation; may not require immediate action.
- Health Update: Provides updated information regarding an incident or situation; unlikely to require immediate action.
- Info Service: Provides general information that is not necessarily considered to be of an emergent nature.
###
This message was distributed to state and local health officers, state and local epidemiologists, state and local laboratory directors, public information officers, HAN coordinators, and clinician organizations.
###

